Medical devices
Our speciality
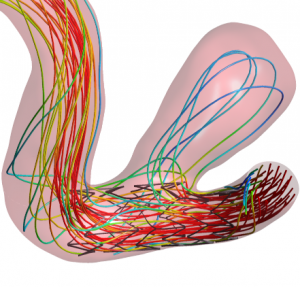
Simulations are an integral part of the development of medical devices. Optimizations can be simulated and carried out even before the test phase begins. This leads to a significant reduction in prototype tests and unavoidable animal testing.In medical technology, the trend is also towards “medical remanufacturing”. This means that one-way becomes reusable, the recycling of plastic medical products is unavoidable for reasons of environmental sustainability and scarcity of resources, requiring targeted product adaptation in terms of dismantlability, repairability and sterilisability. Here, too, the use of targeted simulations is useful.
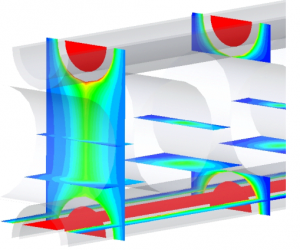
Oxygenators
As part of a heart-lung machine, the oxygenator is designed to ensure optimum gas exchange with minimal blood damage.

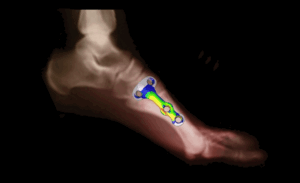
Anesthesia Technology
Numerical flow models can be used to analyze the complex processes involved in vaporization in an emergency anesthesia machine, taking into account the transfer of heat of vaporization and heat conduction in the walls. The pressure rise in the closed
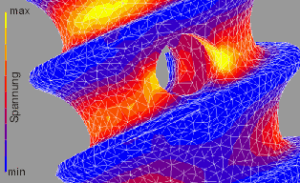
Dental implants
With a length of 8-14 mm and a diameter of 3-5 mm, dental implants must withstand masticatory forces of up to 500 N for decades. Our advanced simulation tools can be used to perform analyses of static strength, life time